|
3/31/2024 0 Comments Periodic table molar mass rounded
1: Moles to Mass Conversion with Elements. According to the periodic table, 1 mol of U has a mass of 238.0 g, so the mass of 2 mol is twice that, or 476.0 g. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. Using the periodic table to find the mass for each mole of our elements we have: 1moleC 1 moleC ×(12.011gC 1 moleC) 12.011gC (8.3.8) (8.3.8) 1 m o l e C 1 m o l e C × ( 12.011 g C 1 m o l e C) 12.011 g C. Please note that the elements do not show their natural relation towards each other as in the Periodic system. First we need to determine the mass of one mole of methane (CH 3 OH). The unity for atomic mass is gram per mol. The lightest chemical element is Hydrogen and the heaviest is Hassium. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight). Do Symbol Hg Boron Carbon 5 6 15 Nitrogen 7 16 Oxygen 8 17 Fluorine 9 4.00 - Neon not round them further when performing calculations. This list contains the 118 elements of chemistry. Helium 2 He 13 14 All average masses are to be treated as measured quantities, and subject to significant figure rules. /PeriodicTableSigFig-NoBG-56a12da75f9b58b7d0bcd00f.png) 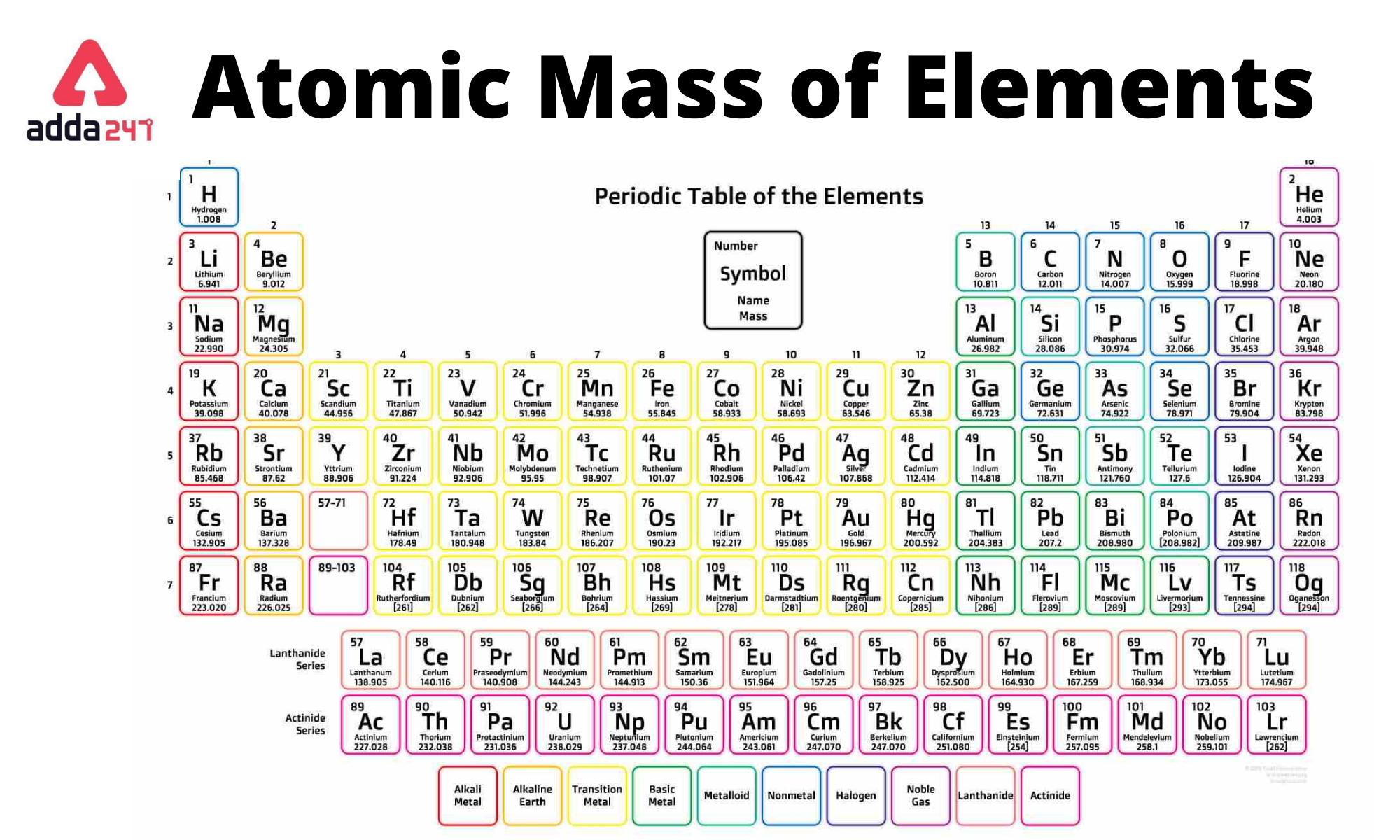
Every hydrogen atom has one proton in its nucleus. Hydrogen, at the upper left of the table, has an atomic number of 1. In this table, an elements atomic number is indicated above the elemental symbol. Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (C 12 H 22 O 11 (sucrose)). Then, lookup atomic weights for each element in periodic table: C: 12.0107, H: 1.00794, O: 15.9994. Separation and Concentration Purification RequestĬhemical elements listed by atomic mass The elements of the periodic table sorted by atomic massĬlick on any element's name for further information on chemical properties, environmental data or health effects. The periodic table (see figure below) displays all of the known elements and is arranged in order of increasing atomic number. First, compute the number of each atom in C 12 H 22 O 11 (sucrose): C: 12, H: 22, O: 11.Plant Inspection & Process Optimalisation.Thus, the molar mass of bilirubin can be expressed as 584.73 g/mol, which is read as “five hundred eighty four point seventy three grams per mole. Atomic Mass of First 30 Elements - Atomic Mass of Elements is Measured with the Help of its Unit - Atomic Mass Unit. The division sign (/) implies “per,” and “1” is implied in the denominator. For example, the molar mass of Ba(OH) 2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: 1 Ba molar mass:īecause molar mass is defined as the mass for 1 mol of a substance, we can refer to molar mass as grams per mole (g/mol). The molar mass of a substance ( M) is the weight of one mole of the substance. Fe4 Fe (CN)63, NaHCO3, ch3coonh4, h2so4, pb (c2h3o2)23h2o, caso41/2h2o) and press Enter or click Calculate button. In formulas with polyatomic ions in parentheses, the subscript outside the parentheses is applied to every atom inside the parentheses. Enter a chemical formula to calculate its molar mass (e.g. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. adenosine (C 10H 13N 5O 4), a component of cell nuclei crucial for cell divisionīe careful when counting atoms.barium sulfate (BaSO 4), used to take X rays of the gastrointestional tract.What is the mass of 1 mol of each substance? \( \newcommand\): Moles to Mass Conversion with Compounds
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
